The 4th annual European bacteriophage future conference hosted by KISACO Research was held on 23-24 November 2021 in Brussels, Belgium. The conference brought together international experts from a wide range of backgrounds in the phage industry, all of whom are interested in phages and phage-related therapeutic applications, the conference will focus on the treatment and application of bacteriophages from a commercial and regulatory perspective.

Diversity of phage applications
The meeting focused primarily on the clinical applications of bacteriophages in humans (phage therapy) , but also covered applications in the animal industry (livestock and aquaculture) .
At present, natural phages have been widely used in clinical treatment, and engineered phages have also begun to be used in academic fields, and have broader application prospects than natural phages. Lyases (active enzymes produced by bacteriophages that hydrolyze bacterial cell walls) have been favoured by companies such as PhagoMed, Bioharmony, Lysando and Micreos, all of which are developing lyase products. But phage applications are not limited to phages themselves and lyases, phage particles can also be utilized as vectors for the delivery of targeted antimicrobials (e.g. , Eligo Bioscience utilizes the CRISPR-Cas9 system to selectively shear antibiotic resistance or virulence genes carried in bacteria, among others) .
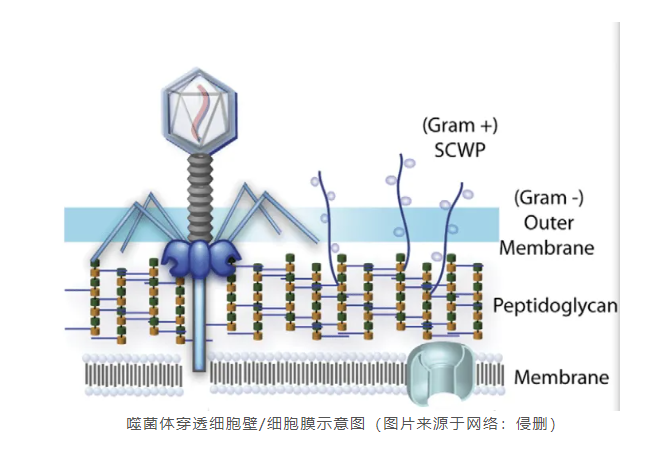
Matching bacteriophages to bacteria
Selection of specific phages to combat specific bacterial strains is critical for phage therapy. How to improve the speed and accuracy of phage screening was discussed. Dr. Ronen Hazan (Principal Investigator, Jerusalem University, Israel) , Dr. Bob Blasdel (Research Director, Vesale Pharmaceuticals) , and Dr. Cindy Fevre (r & D Director, Pherecydes Pharma) presented different phage screening experiences and their preferred approaches. Dr. Hazan noted the importance of a comprehensive test that combines plaque and kinetic results for screening. Dr Fevre also prefers to use both methods to screen phages, especially when they are used to treat patients.
In addition, Dr. Blasdel presented the high-throughput screening phage approach adopted by Vésale Pharma to meet the need of hospitals and research institutions to select the best phage for patient use in a short period of time. This method is based on the luciferase luminescence technology to detect the amount of ATP released after the bacteriophage lyses the bacteria, and the ATP is linearly related to the fluorescence intensity in a certain range, therefore, ATP release can be calculated by measuring the fluorescence intensity, and lytic phages can be screened out. However, the reliability of this method depends on the previous determination of a large number of different phages. Therefore, to test phages as much as possible in combination with kinetic detection.
Finally, Dr. Natalie Ma, co-founder of Felix Biotechnology, describes the possibility of accelerating phage-bacteria matching in the future. Felix Biotechnology feeds host bacteria range data to artificial intelligence algorithms to find the sequences of genes phages use to infect bacteria.
Challenges in phage manufacturing
Phage preparation process, GMP production from small-scale to large-scale is also the main topic of the conference. Dr. Barbara Hubad, Jafral’s chief operating officer, shared her experience with the mass production of GMP phages. According to Dr Hubad, the most important aspect of GMP production is the prevention of phage cross-contamination, particularly when using different phages to make cocktails. Another challenge is that the stability of phages may also change as they scale up from small to large.
Dr. Danish Malik, from Loughborough University, UK, describes work on process optimization for mass production of high titer-purified phages. For example, he says, continuous production of phages in relatively small volumes can improve yields. Dry powder formulations can theoretically meet the stability requirements of all phages, although the stability of phages varies.
Dr. Adam Ostrowski, an expert in CELLEXUS technology applications, advocates the introduction of disposable devices for phage production. Cellexus has developed a Bioreactor system that provides rapid and easy phage production and minimises cross-contamination by using disposable filter bags. Due to the challenges faced by phage GMP production, the industry has paid increasing attention to phages and continues to seek solutions for phage scientists.
“Fixed cocktail” and“Individuation”
Phage applications often face a debate over whether to use a“Fixed cocktail” or an“Individualised” phage therapy. Dr. Bob Blasdel supports the creation of small, individually characterized phage libraries that can then be selected as active drug ingredients and assembled into cocktails. In addition, feedback from bacterial susceptibility and effectiveness data from ‘clinical trials’ can inform models and improve phage selection criteria.
Overall, the current consensus seems to be that the two approaches are not mutually exclusive but complementary. Clinical trials tend to use the immobile cocktail method, whereas in the future, one or more phages taken from a phage library for personalized therapy may be a more promising regimen.
The importance of clinical trials
Representatives of the health authorities at the conference, Peter Beyer (Senior Advisor to the World Health Organization) and Dr. Cara Fiore (microbiologist and senior regulatory reviewer at the Food and Drug Administration) , highlighted the need to conduct clinical trials, to assess the safety and effectiveness of phage therapy.
Dr. Shawna McCallin (clinical coordinator, Faculty of Medicine, University of balgrist, Switzerland) provides an objective overview of current clinical trials in the field of phage therapy. So far, 26 phage studies have been registered, of which six are actively recruiting and five have begun — a record for phage therapy. Success in Phase II trials is a critical step for phage therapy, she says, because three of the four phase II studies to date have failed. She collaborated with Dr Sarah Djabara, an infectious disease physician at Queen Astrid Military Hospital in Belgium, to create an international database of phage therapies. This international initiative aims to collect data in a centralized and standardized manner to increase transparency and reduce subjective bias in reporting.
The phage therapy of the developing world
Antimicrobial resistance (Amr) is rampant and is estimated to kill more than 10m people by 2050. African and Asian countries will pay a heavy price for the lack of medicines.
Dr. Tobi Nagel, founder and president of Phages for Global Health, described her work at a nonprofit philanthropic organization that promotes the use of Phages in Africa and Asia. Because of the AMR crisis, there is a need to avoid the delays in drug delivery seen in the AIDS and covid-19 epidemics, and one way to achieve this is for developing countries to produce drugs for their own populations. To this end, she has promoted the establishment of phage libraries in developing countries with the support of international and public philanthropic foundations, the first step in establishing the phage library was to organize seminars on the application of phage isolation in African countries such as Uganda, Kenya, Ghana and Tanzania. More than 1,200 scientists were trained in phage biology, 450 phages were isolated across the continent, and 50 phage projects were created.

Global annual deaths due to antimicrobial resistance predicted by 2050
The future of the phage field
What are the prospects for phage therapy in five years?
With a growing awareness of antibiotic resistance and a dearth of future antibiotics, the phage therapy is seen as having a bright future. For Dr Hessel, precise targeting of microbes holds great promise and requires further study. Dr Pirnay, on the other hand, envisions a more distant future in which cell-free, just-in-time production of phages is possible. Finally, DRS Corsini and McCallin insist on the importance of clinical trials, as the number of ongoing clinical trials is greater than ever before and may provide a clearer understanding of the potential of phage therapy.
Conclusion
In conclusion, the 2021 European future of bacteriophages conference confirmed a trend observed over the past few years: the increasing international popularity of phage applications, the start of more clinical trials, and the emergence of a new generation of bacteriophages, the company introduced more new products and technologies. The safety of phage therapy is now well established, and the biggest challenge in the future remains to prove its effectiveness. In the treatment of bacterial infections, approaches such as phages, lyases, or phage-derived antimicrobials should not be excluded but should be added to our process of combating antimicrobial resistance.


-300x169.png)
